
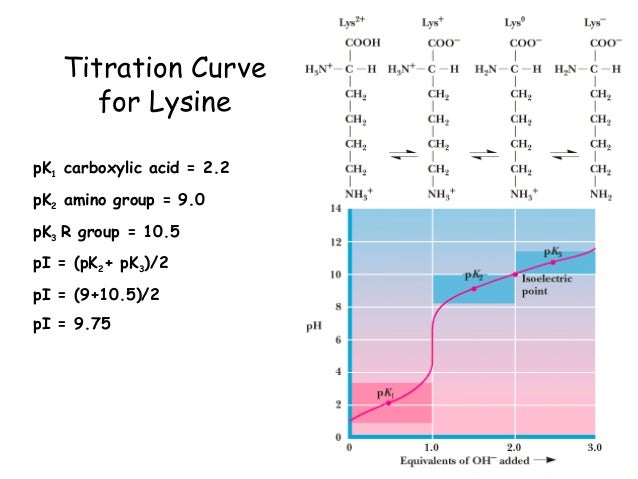
For neutral amino acids, the side chains are neutral and the isoelectric point is given simply by the average of the pK a values of carboxylic acid and amine.

In case of proteins isoelectric point mostly depends on seven charged amino acids: glutamate (-carboxyl group), aspartate (-carboxyl group), cysteine (thiol group), tyrosine (phenol group), histidine (imidazole side chains), lysine (-ammonium group) and arginine (guanidinium group). threonine-, & lysine - enriched pTHRESH Impact phenomena in threshing affected by energy. Isoelectric point definition: Isoelectric point (pI) is a pH in which net charge of protein is zero. For amino acids that have no ionizable side chain, the pI value is the. Note: The isoelectric point is given by the average of the pK a values that involve the zwitterions, not just by the pK a values that describe the carboxylic acid group and the amine group. ( equieconomic evaluation of thinning artificial Pi. Isoelectric point is the pH of an amino acid at which it has no net electric charge. Since the isoelectric point is given by the average of the pK a values that involve the zwitterion, so we can write the formula for lysine as: the net charge on the nitrogen-containing groups is +1 and balances the charge of -1 on the alpha-carboxyl group.

